The Gut As a Brain
The Gut... As a Brain?
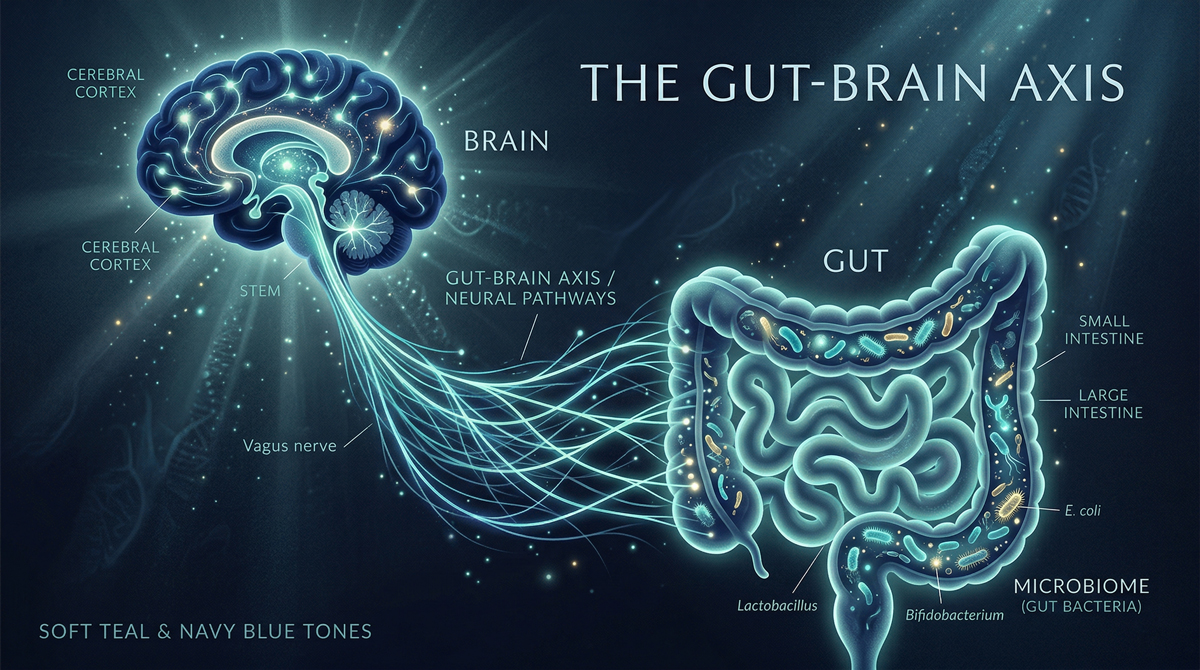
The gut is far more than a tube for digestion. It contains its own nervous system, dense immune tissue and a complex microbial ecosystem, forming an integrated gut-brain-immune axis that many authors now describe as a functional second brain (Furness, 2012; Carabotti et al., 2015).
The enteric nervous system (ENS) is embedded in the wall of the gastrointestinal tract and comprises sensory neurones, interneurones and motor neurones capable of coordinating motility, secretion and local blood flow independently of the central nervous system (Furness, 2012). Because the ENS can generate complex reflex patterns on its own and communicates bidirectionally with the brain via vagal and spinal pathways, it is often referred to as the body's second brain (Furness, 2012; Mayer et al., 2015).
Stress, the Brain-Gut Axis and Gut Electrical Activity
Acute and chronic psychological stress activate the hypothalamic-pituitary-adrenal axis and autonomic nervous system, with clear downstream effects on gastrointestinal function (Konturek et al., 2011). Experimental and translational studies show that stress can disrupt intestinal slow-wave electrical activity and peristaltic patterns, leading to dysmotility, visceral hypersensitivity and barrier changes, via ENS circuitry and microbiota-dependent signalling (Tache and Brunnhuber, 2008; Konturek et al., 2011).
Microbiome, Sugar and Gut Health
The intestinal microbiome is a dense community of bacteria, archaea, fungi and viruses that contributes to digestion, produces bioactive metabolites, educates the immune system and maintains epithelial barrier integrity (Belkaid and Hand, 2014; Zheng et al., 2020).
Diet is one of the strongest modulators of this system. In animal models, high-glucose or high-fructose diets reduce microbial diversity, decrease Bacteroidetes, increase Proteobacteria and induce low-grade inflammation, increased intestinal permeability and metabolic endotoxaemia, even in the absence of major weight gain (Do et al., 2018; Nobs et al., 2019).
Gut, Immunity and Intolerances
Approximately 70% of immune cells are located in gut-associated lymphoid tissue, where continuous sampling of luminal contents underpins tolerance to food and commensals versus defence against pathogens (Belkaid and Hand, 2014; Zheng et al., 2020).
When the microbiota is disturbed and the epithelial barrier becomes more permeable, larger quantities of dietary antigens, toxins and microbial products can cross into the lamina propria and systemic circulation, driving inappropriate immune activation and low-grade inflammation (Fukui, 2016; Bischoff et al., 2014).
Leaky Gut and Intestinal Permeability
The popular notion of leaky gut corresponds to increased intestinal permeability, usually via disruption of tight-junction proteins that link epithelial cells (Bischoff et al., 2014; Camilleri, 2019). Experimental and clinical work indicates that high-sugar, high-fat and ultra-processed diets, as well as certain emulsifiers and food additives, can impair tight-junction integrity, thin the mucus layer and alter the microbiota in ways that favour barrier breakdown and mucosal inflammation (Fukui, 2016; Agus et al., 2021).
Stool Tests and SIBO Breath Tests: Useful, With Caveats
Stool microbiome panels can characterise faecal microbial composition and are valuable in research and in specific clinical scenarios (Marchesi et al., 2016; Zheng et al., 2020). However, expert reviews emphasise that faecal data are only a proxy for mucosa-associated communities and do not by themselves diagnose specific diseases (Marchesi et al., 2016).
Hydrogen-methane breath testing is the most widely used non-invasive tool for assessing small intestinal bacterial overgrowth (SIBO) and intestinal methanogen overgrowth (Rezaie et al., 2017). Both stool microbiome analyses and SIBO breath tests can illuminate aspects of the gut-brain-immune axis but their findings should always be integrated with clinical history, symptoms, diet, medications and co-morbidities (Marchesi et al., 2016; Rezaie et al., 2017).
Sources
Agus, A. et al. (2021) Cell Host and Microbe, 29(6). | Belkaid, Y. and Hand, T.W. (2014) Cell, 157(1). | Bischoff, S.C. et al. (2014) BMC Gastroenterology, 14. | Camilleri, M. (2019) Gut, 68(8). | Carabotti, M. et al. (2015) Annals of Gastroenterology, 28(2). | Do, M.H. et al. (2018) Nutrition, 54. | Fasano, A. (2020) Physiological Reviews, 100(1). | Fukui, H. (2016) Inflammation and Regeneration, 36. | Furness, J.B. (2012) Nature Reviews Gastroenterology and Hepatology, 9(5). | Konturek, P.C. et al. (2011) Journal of Physiology and Pharmacology, 62(6). | Marchesi, J.R. et al. (2016) Gut, 65(2). | Mayer, E.A. et al. (2015) Journal of Clinical Investigation, 125(3). | Moloney, R.D. et al. (2016) CNS Neuroscience and Therapeutics, 22(2). | Nobs, S.P. et al. (2019) Cell Metabolism, 30(3). | Rezaie, A. et al. (2017) American Journal of Gastroenterology, 112(5). | Zheng, D. et al. (2020) Cell Research, 30(6).
Join Our Wellness Community
Subscribe for the latest insights on aesthetics, health tips, and exclusive offers from Eden Clinic.



